|
Although the Bohr model has been supplanted by other models, its underlying principles remain valid. Das Atommodell von BOHR, so wie es oft genau wie in der Animation in Abb. Bohr developed the Bohr model of the atom, in which he proposed that energy levels of electrons are discrete and that the electrons revolve in stable orbits around the atomic nucleus but can jump from one energy level (or orbit) to another. According to his model for a diatomic molecule, the electrons of the atoms of the molecule form a rotating ring whose plane is perpendicular to the axis of the molecule and equidistant from the atomic nuclei. 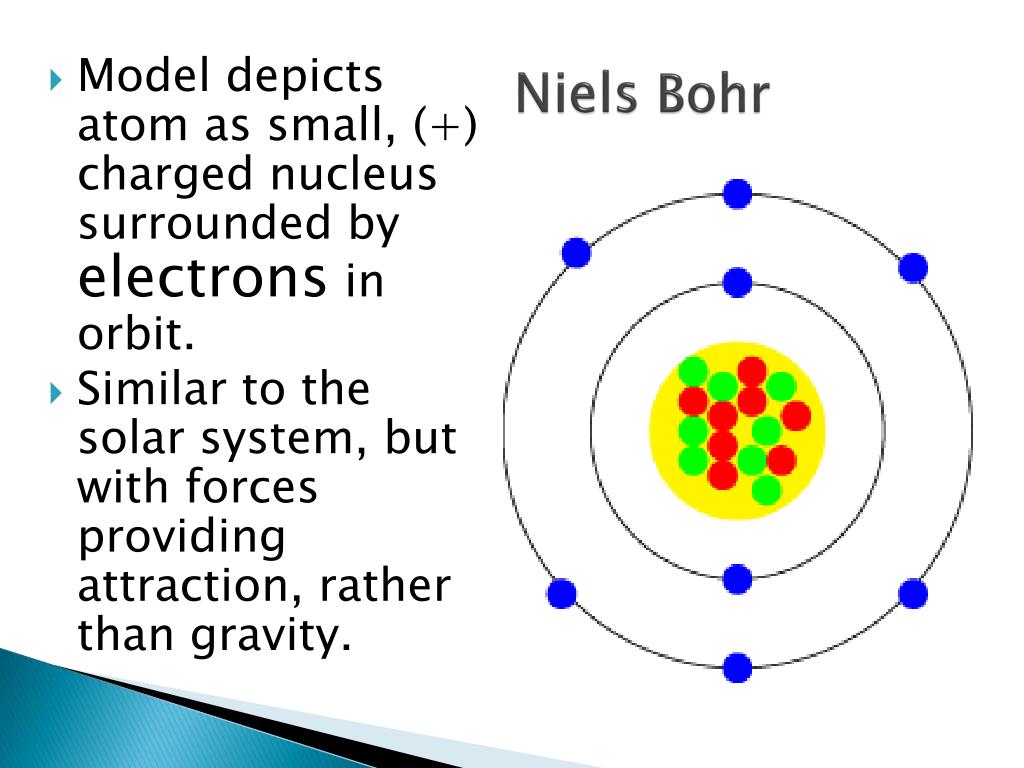
Because a hydrogen atom with its one electron in this orbit has the lowest possible energy, this is the ground state (the most stable arrangement of electrons for an element or a compound), the most stable arrangement for a hydrogen atom. 1 Atommodell von BOHR mit den auf diskreten Bahnen um den positiv geladenen Kern kreisenden Elektronen. Niels Bohr proposed a model of the atom and a model of the chemical bond. 
\) indicates that the electron-nucleus pair is more tightly bound when they are near each other than when they are far apart. Bohr model, description of the structure of atoms proposed in 1913 by the Danish physicist Niels Bohr.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
